 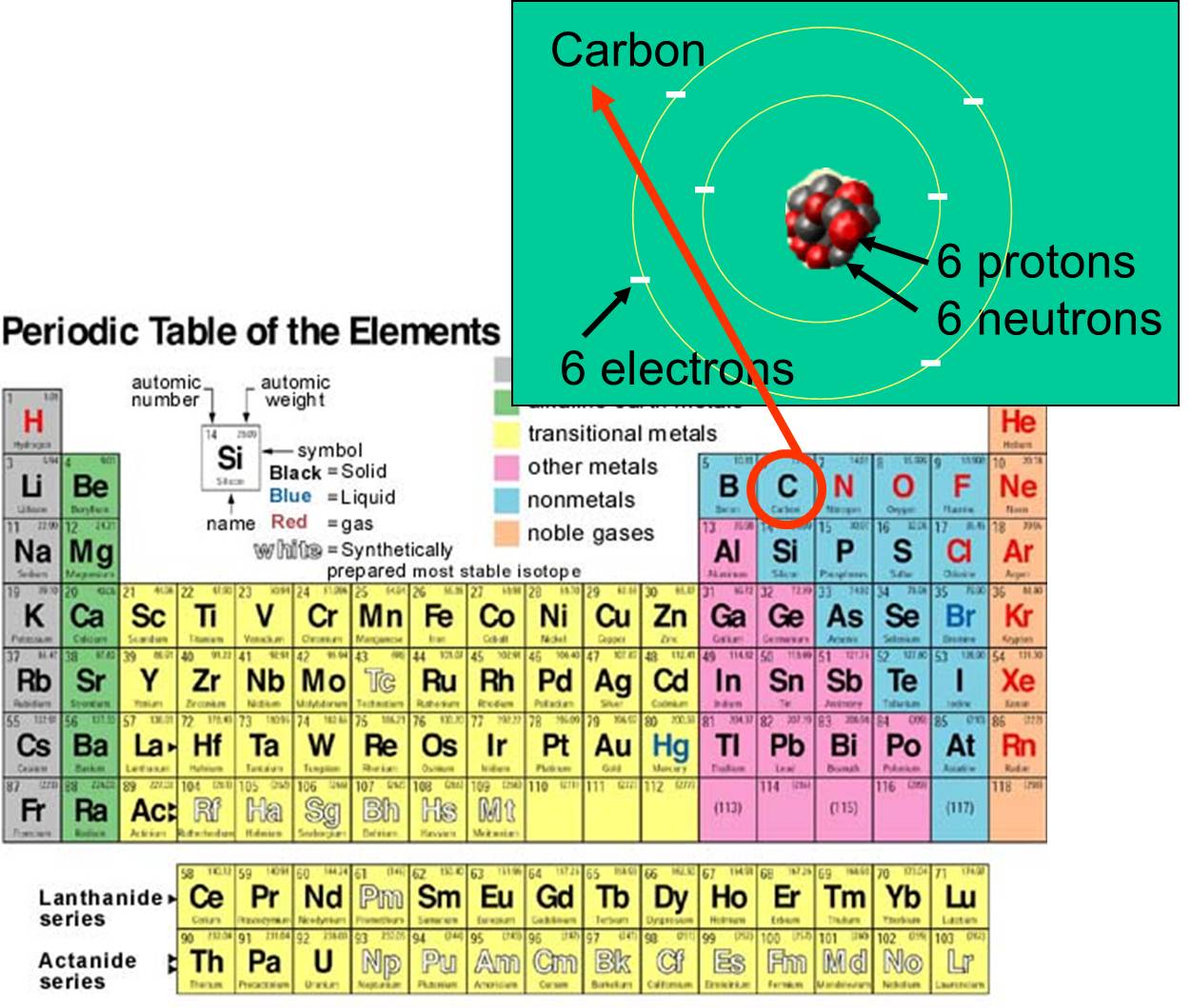
Pure substance chemical properties What is not a pure substance? C 12 H 22 O 11 , and a pure substance that is a compound but not considered a molecule is sodium chloride, N a C l NaCl N a Cl (table salt). This is a partial list of physical properties that can be observed, measured, and predicted for pure substances under normal conditions (room temperature and one earth atmosphere of pressure): Sodium chloride from the Dead Sea is physically the same as sodium chloride mined in Kansas. The periodic table is a tabular arrangement of the chemical elements by increasing atomic number which displays the elements so that one may see trends in their properties. Pure substances have a constant composition. Chemical elements are identified by the number of protons in the nuclei of their atoms, 1 known as the element's atomic number. The basic particle that constitutes a chemical element is the atom. No matter where a given pure substance is found, it will have identical physical properties to every other pure substance of the same chemical makeup. A chemical element is a chemical substance that cannot be broken down into other substances by chemical reactions. In other words, each atom has the same number of electron shells. Elements in the same period all have the same highest unexcited electron energy level or same ground state energy level. Interactive periodic table with up-to-date element property data collected from authoritative sources. Atoms or ions with the same electronic configurations are said to be. Isoelectronic chemical species typically display similar chemical properties. The term means 'equal electric' or 'equal charge'. These predictable properties make pure substances the foundation of chemistry because known interactions can be used to form new materials and new types of matter. In chemistry, the term period refers to a horizontal row of the periodic table. Isoelectronic refers to two atoms, ions, or molecules that have the same electronic structure and the same number of valence electrons. Whether element, molecule, or compound, pure substances share similar physical and chemical properties. As you more down the periodic table, there are more elements per element period. Classes of the periodic table 1.6 LIST the. Classes of the periodic table 1.5 Given a periodic table, IDENTIFY the following subdivisions: a. In other words, each atom has the same number of electron shells. 1.4 DEFINE the following subdivisions of the periodic table: a. Magnesium, for example, is placed in the alkali earths column, with other elements whose reactions are similar: Mendeleev realized that the table in front of him lay at the very heart of chemistry. Elements in the same period all have the same highest unexcited electron energy level or same ground state energy level. Similar chemical properties to other elements in the same column - in other words similar chemical reactions. Pure substance definition Properties of pure substances In chemistry, the term period refers to a horizontal row of the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
